Bringing a medical device to the U.S. market is a journey of innovation, but it is also one of strict regulatory adherence. For most moderate-risk devices, the 510(k) Premarket Notification is the vital bridge between a concept and a commercially available product.
At Satori One Click Solutions LLP, we understand that the complexity of the Food and Drug Administration (FDA) requirements can be overwhelming. This detailed guide breaks down the 510(k) submission process into manageable steps, ensuring your business stays compliant while reaching its growth milestones.
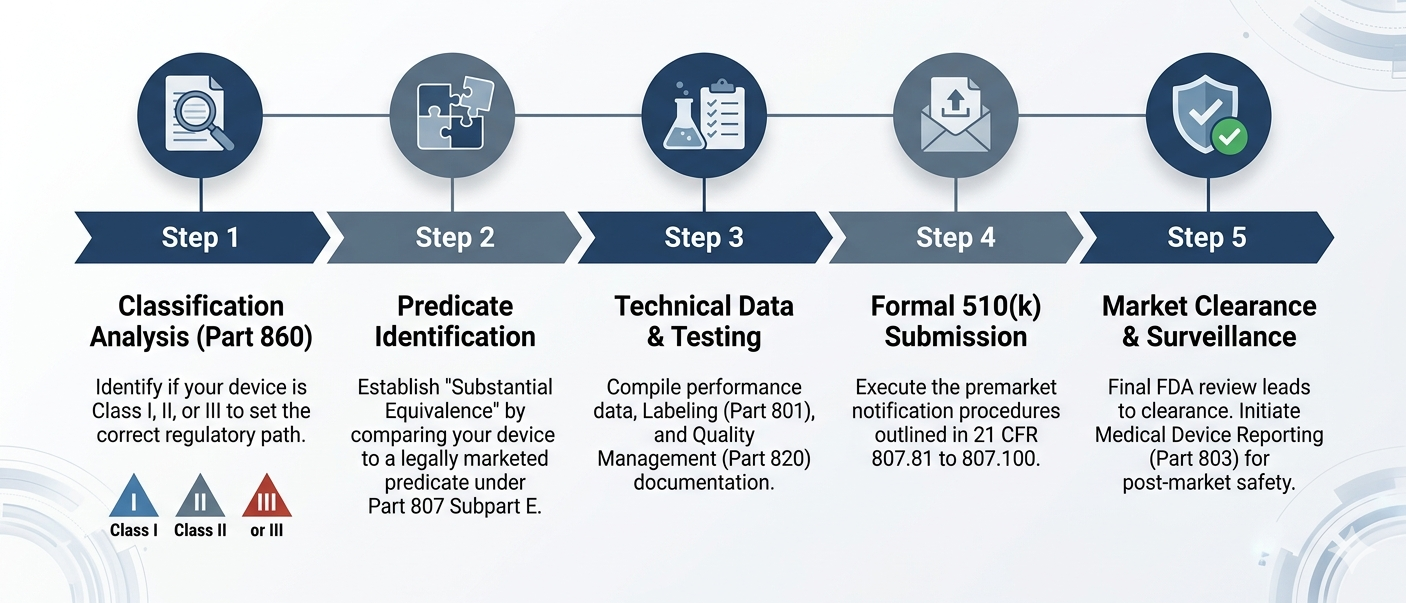
Step 1: Determine Your Device Classification (Part 860)
The first step in any regulatory strategy is identifying the "class" of your device. Under 21 CFR Part 860, the FDA categorizes medical devices into Class I, II, or III based on the level of control necessary to ensure safety and effectiveness.
- Class I: Low risk (e.g., bandages).
- Class II: Moderate risk (e.g., infusion pumps). Most 510(k) submissions fall into this category.
- Class III: High risk or life-sustaining (e.g., replacement heart valves), which often require a more rigorous Premarket Approval (PMA),.
Correct classification is the foundation of your submission. Satori One Click Solutions provides expert analysis to ensure you aren't over-complicating your path by misclassifying your device.
Step 2: Identify a Predicate Device and Substantial Equivalence
A 510(k) is a submission made to the FDA to demonstrate that a new device is "substantially equivalent" to a device already legally marketed in the U.S. that does not require a PMA. This already-marketed device is known as the predicate device.
- Your device must have the same intended use as the predicate.
- It must have the same technological characteristics, or if they differ, the differences must not raise new questions of safety and effectiveness.
Step 3: Compile the Submission (Part 807 Subpart E)
The formal procedures for your submission are governed by 21 CFR Part 807 Subpart E (sections 807.81 – 807.100). A successful submission is not just about data; it is about organization.
Key components of your eSTAR file include:
- Labeling (Part 801): You must include proposed labels and directions for use that meet federal standards.
- Quality Management (Part 820): Evidence that your manufacturing processes comply with the Quality Management Systems Regulation.
- Performance Testing: Bench testing or clinical data showing your device performs as intended.
Step 4: Post-Market Readiness and Surveillance
Clearance is not the end of the road. To maintain your market position, you must have systems in place for:
- Medical Device Reporting (Part 803): Procedures for reporting adverse events or malfunctions.
- Post-market Surveillance (Part 822): Plans for monitoring the device's performance once it is in use.
- Correction and Removals (Part 806): Protocols for managing potential recalls.
Why Partner with Satori One Click Solutions LLP?
The regulatory landscape is constantly evolving. Attempting to navigate Establishment Registration (Part 807) or complex Classification Procedures (Part 860) alone can lead to costly delays.
Satori One Click Solutions LLP provides a "one-click" experience for B2B clients, offering:
- Expert Consultation: We help you anticipate the FDA's questions before they ask them.
- Streamlined Documentation: We ensure your files are "people-first," unique, and up-to-date qualities that satisfy both FDA reviewers.
- End-to-End Support: From initial classification to post-market safety alerts, we are your long-term compliance partner.
Get in touch with us today and take the first step toward FDA clearance:
Email: satoriocs@gmail.com
Website: satoriocs.com
Phone: +91-9829098077 / 9216598077